24-hour hotline:+8613662168047
Keyword search: battery plant , lithium battery factory , power bank works , lifepo4 battery mill , Pallet Trucks LiFePO4 Battery, LiFePO4 Pallet Trucks Battery, Lithium Pallet Trucks Battery,
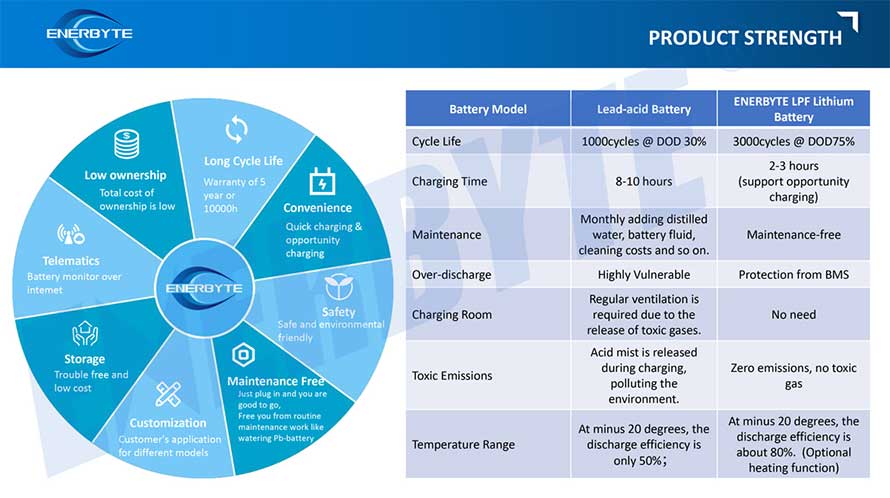
Lithium iron batteries are a type of battery in the lithium battery family, with the main positive electrode material being lithium iron phosphate material, also known as lithium iron batteries. Compared with traditional lead-acid batteries, lithium-ion batteries have significant advantages in operating voltage, energy density, and cycle life.
Compared with traditional lead-acid batteries, lithium iron batteries have the following advantages: high energy density, strong safety, good high-temperature performance, high power output, long cycle life, light weight, saving costs for strengthening computer rooms, small size, long battery life, and good safety.
In 1990, Sony Corporation was the first to launch lithium-ion batteries using LiCoO2 as the positive electrode material in the laboratory, and began industrial production in 1991. Compared with traditional lead-acid batteries, lithium-ion batteries have significant advantages in operating voltage, energy density, and cycle life. So, in the past two decades, lithium-ion batteries have been widely used in portable electronic devices, power tools, and other fields. In recent years, with the global attention to energy conservation and emission reduction, lithium-ion batteries have gradually been applied in various industries such as communication, national power grid, and electric vehicles. For energy conservation and emission reduction in the communication power supply industry, it is required that batteries have smaller volume, lighter weight, longer lifespan, higher temperature resistance, easier maintenance, more stable performance, and are more environmentally friendly. Therefore, in order to meet these needs, lithium-ion batteries are gradually shifting towards high-capacity batteries, and lithium iron phosphate batteries for communication have emerged. ABC lithium iron battery is a type of battery in the lithium battery family, with the main positive electrode material being lithium iron phosphate material, also known as lithium iron battery.
Lithium iron batteries are not as safe as some friends online claim. There is also a risk of explosion. Technology requires honesty and rigor.
The lithium iron phosphate battery 26650-3AH was overcharged at 3C10V, resulting in an explosion of the battery. Repeated testing yielded similar results.
(Note that this experiment was conducted on an unformed product, and the narrator cannot provide photos)
When lithium batteries are charged and discharged with high current, the internal temperature of the battery continues to rise, and the gas generated during the activation process expands, increasing the internal pressure of the battery. When the pressure reaches a certain level, if there are scars on the outer shell, it will rupture, causing leakage, fire, and even explosion. So, everyone must pay attention to safety when using it.
However, in normal use, the battery management system is generally used to protect the power lithium battery, so there is almost no explosion phenomenon. However, for mobile phone batteries, due to inadequate protective measures, they are more prone to explosions.
The full name of lithium iron battery is lithium iron phosphate lithium-ion battery. Due to its performance being particularly suitable for power applications, some people also call it "lithium iron power battery". (hereinafter referred to as "Lithium Iron Battery")
Working principle of lithium iron battery (LiFePO4)
The internal structure of LiFePO4 battery is shown in Figure 1. On the left is LiFePO4 with an olivine structure as the positive electrode of the battery, connected to the positive electrode by aluminum foil, with a polymer separator in the middle that separates the positive and negative electrodes. However, lithium ions Li+can pass through while electrons e - cannot. On the right is the negative electrode of the battery composed of carbon (graphite), connected to the negative electrode of the battery by copper foil. The electrolyte of the battery is located between the upper and lower ends, and the battery is enclosed by a metal shell.
During charging of LiFePO4 batteries, the lithium ions Li+in the positive electrode migrate to the negative electrode through the polymer separator; During the discharge process, lithium ions Li+in the negative electrode migrate to the positive electrode through the separator. Lithium ion batteries are named after the back and forth movement of lithium ions during charging and discharging.
Main performance of LiFePO4 battery
The nominal voltage of LiFePO4 battery is 3.2V, the termination charging voltage is 3.6V, and the termination discharging voltage is 2.0V. Due to differences in the quality and process of positive and negative electrode materials and electrolyte materials used by various manufacturers, there may be some differences in their performance. For example, for the same model (standard battery packaged in the same way), there is a significant difference in battery capacity (10% to 20%).
Lithium iron phosphate batteries are actually lithium-ion batteries that use lithium iron phosphate as the positive electrode material. As for lithium-ion batteries, there are various types of positive electrode materials such as lithium cobalt oxide, lithium manganese oxide, lithium nickel oxide, ternary materials, lithium iron phosphate, etc. Among them, lithium iron phosphate is the most commonly used material in the lithium battery industry today.
Lithium iron phosphate batteries are connected to the positive electrode of the battery by aluminum foil. On the left is a polymer barrier that separates the positive and negative electrodes, but the lithium ion Li can start and end while the electron e - cannot. On the right is the negative electrode of the battery composed of carbon, which is connected to the negative electrode of the battery by copper foil. The electrolyte of the battery is between the upper and lower ends of the battery, and the battery is enclosed by a metal shell. When LiFePO4 batteries are charged, the lithium ion Li in the positive electrode moves from the polymer barrier to the negative electrode; During the discharge process, the lithium ions Li in the negative electrode migrate towards the positive electrode, and the lithium-ion battery is named after the back and forth migration of lithium ions during charging and discharging.
The working principle of lithium-ion batteries is that during battery charging, Li migrates from the 010 surface of the lithium iron phosphate crystal to the surface of the crystal. Under the effect of electric field force, it enters the electrolyte, passes through the barrier, and then migrates to the surface of the graphite crystal through the electrolyte, and then embeds into the graphite lattice. After lithium ions are deintercalated from lithium iron phosphate, lithium iron phosphate is converted into iron phosphate; When the battery is discharged, Li is deintercalated from the graphite crystal, enters the electrolyte, passes through the barrier, and then moves to the surface of the lithium iron phosphate crystal through the electrolyte. Then, it is re embedded into the lattice of lithium iron phosphate through the 010 surface, and flows through the conductor to the positive electrode of lithium iron phosphate to start discharging.
For the conductivity of the positive and negative electrodes of lithium iron phosphate batteries, conductive agents must be added to the positive and negative electrodes of the battery to form an active component in the battery. In the process of theoretical description and production, it is necessary to describe a series of experiments to verify the above three equations, establish mathematical models or historical formulas, and then use these models or formulas to describe lithium-ion batteries from beginning to end.
At that time, the domestic lithium-ion battery technology was so advanced that it had a world leading level. The industry still focused on lithium-ion batteries with lithium iron phosphate as the positive electrode, and many countries developed new lithium-ion battery materials. However, there was no large-scale production at that time. Some scholars pointed out that for at least 10 years, lithium iron phosphate positive electrode materials were the mainstream industry that opened up.
Lithium batteries are a type of battery that uses lithium metal or lithium alloy as the negative electrode material and a non-aqueous electrolyte solution, while lead-acid batteries are a type of battery where the electrode is mainly made of lead and its oxides, and the electrolyte is a sulfuric acid solution.
1、 Lithium battery
1. Basic Introduction
Lithium battery refers to a battery that contains lithium (including metallic lithium, lithium alloys, lithium ions, and lithium polymers) in an electrochemical system. Lithium batteries can be roughly divided into two categories: lithium metal batteries and lithium-ion batteries. Lithium ion batteries do not contain metallic lithium and are rechargeable. The fifth generation product of rechargeable batteries, lithium metal batteries, was born in 1996. Its safety, specific capacity, self discharge rate, and cost-effectiveness are all superior to lithium-ion batteries. Due to their own high-tech requirements, only a few companies in certain countries are currently producing this type of lithium metal battery.
2. Working principle
(1) Lithium metal battery:
Lithium metal batteries generally use manganese dioxide as the positive electrode material, metallic lithium or its alloy metal as the negative electrode material, and non-aqueous electrolyte solutions.
Discharge reaction: Li+MnO2=LiMnO2
(2) Lithium ion battery:
Lithium ion batteries generally use lithium alloy metal oxides as positive electrode materials, graphite as negative electrode materials, and non-aqueous electrolytes.
The reaction occurring on the charging positive electrode is
LiCoO2==Li (1-x) CoO2+XLi++Xe - (electron)
The reaction that occurs on the charging negative electrode is
6C+XLi+Xe -=LixC6
Total reaction of rechargeable battery: LiCoO2+6C=Li (1-x) CoO2+LixC6
2、 Lead acid battery
1. Basic Introduction
After nearly 150 years of development, lead-acid batteries have made significant progress in theoretical research, product types and varieties, electrical performance, and other aspects. Whether in transportation, communication, power, special industries, navigation, or various economic fields, lead-acid batteries play an indispensable and important role. According to the differences in structure and usage of lead-acid batteries, they can be roughly divided into four categories: (1) lead-acid batteries for starting; (2) Lead acid batteries for power use; (3) Fixed valve regulated sealed lead-acid battery; (4) Other categories include small valve regulated sealed lead-acid batteries, lead-acid batteries for mining lamps, etc.
2. Working principle
Valve regulated lead-acid battery charging is the process of connecting an external DC power source to the battery for charging, converting electrical energy into chemical energy and storing it. Discharge is the release of electrical energy from a battery to drive external devices.
When the charging of lead-acid batteries reaches its peak, the charging current is only used to decompose the water in the electrolyte. At this time, the positive electrode of the battery produces oxygen, and the negative electrode produces hydrogen. The gas will overflow from the battery, causing a decrease in electrolyte and requiring periodic water addition.
On the other hand, at the end of charging or under overcharge conditions, the charging energy is used to decompose water, and the oxygen produced by the positive electrode reacts with the sponge like lead in the negative electrode, causing a part of the negative electrode to be in an unfilled state and suppressing the production of hydrogen gas in the negative electrode.

Lithium Batteries ,Ensure Quality
Our lithium battery production line has a complete and scientific quality management system
Ensure the product quality of lithium batteries

Years of experience in producing lithium batteries
Focus on the production of lithium batteries

WE PROMISE TO MAKE EVERY LITHIUM BATTERY WELL
We have a comprehensive explanation of lithium batteries

QUALIFICATION CERTIFICATE
THE QUALITY OF COMPLIANCE PROVIDES GUARANTEE FOR CUSTOMERS
MULTIPLE QUALIFICATION CERTIFICATES TO ENSURE STABLE PRODUCT QUALITY
Providing customers with professional and assured products is the guarantee of our continuous progress.




Applicable brands of our products


 Service hotline
Service hotline